UPDATE: Following the release of this blog post, the maintenance fee due in January 2023 was increased to $4,875 per product.
On Thursday, December 29, 2022, the Pesticide Registration Improvement Act of 2022 (PRIA 5) was signed into law as part of the Consolidated Appropriations Act, 2023. The passing of this bill confirmed PRIA reauthorization and continuation of the protections and resources it provides to both EPA and the pesticide industry through September 30, 2027. The Consolidated Appropriations Act, 2023 also included additional appropriations for the EPA Office of Pesticide Programs, which will provide much needed additional resources.
This is a huge win for EPA and industry. We extend our gratitude to the Agency and PRIA coalition for their efforts towards this reauthorization.
The SRC Team summarized the key changes in PRIA 5 affecting pesticide registrants.
Maintenance Fee Increases
Maintenance fees were set to $4,875 $3,400 per product for 2023. Maximum pesticide registration maintenance fees will increase for fiscal year 2023-2027 as follows:
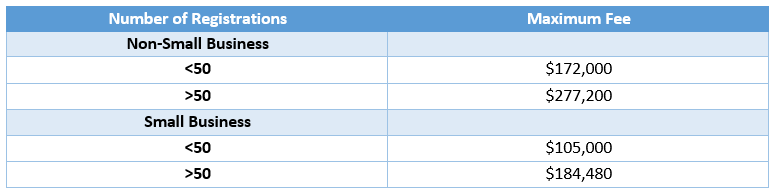
The fee changes are effective retroactive to 10/01/22, which means a supplemental maintenance fee will be due for the 2023 calendar year, though the value and timing is yet to be announced.
Pesticide Registration Services Fees
PRIA 5 includes updated registration fee tables which increase fees by an average of 30%, as well as lengthens the review time for many application categories. The new fees and timelines will take effect on February 27, 2023, 60 days after the bill was signed into law. Applications submitted before this date are still subject to PRIA 4. Please contact your SRC Consultant as soon as possible if you have an action eligible for submission before PRIA 5 takes effect so we can plan to submit within the 60-day window.
Updated PRIA tables can be found here.
Increased Communication and Transparency
The PRIA reauthorization also includes efforts to increase transparency between the Agency Office of Pesticide Programs (OPP) and Industry. Notable provisions include:
- OPP to supply applicants with a justification to extend a PRIA due date and provide the number of days needed to make a decision.
- OPP to release data evaluation records (DERs) to registrants at decision review time.
- During the technical screen an OPP reviewer may determine if a different PRIA fee and review timeline should apply to an application.
- Adds annual reporting requirements on Agency staffing, resource allocation, completed actions, and renegotiations. This report must be published within 120 days of the end of each fiscal year.
- OPP to create a tracking system to provide applicants access to real-time data/status on the progress of their submissions. EPA has 1 year to implement this software.
- OPP staff to be provided improved access to regulatory decision-making tools and data.
- EPA staff compelled to help registrants determine the appropriate fee category for applications that may fall under multiple categories to avoid applicants paying multiple PRIA fees for one application.
- Provides funding and deadlines for EPA to finalize guidance for compliance with the Endangered Species Act for pesticides with outdoor uses.
EPA Process and Workforce Assessments
PRIA 5 provides funding and a 2-year deadline for EPA to hire an outside contractor to conduct a workforce assessment and provide recommendations relating to PRIA 5. EPA will also be granted a 1-year deadline to hire an outside contractor to conduct a process assessment and provide recommendations related to the implementation of PRIA 5.
Reregistration and Expedited Processing Fund
EPA will also, under PRIA 5, set aside the following amounts annually from the Reregistration and Expedited Processing Fund.
- 1/8th of available funds for staffing and resources to process non-PRIA actions. No deadline is included for the processing of these actions. This will hopefully assist with the current backlog and improve timely processing of these equally important actions.
- Up to $500,000 for the development of efficacy test methods for antimicrobial pesticide devices making public health claims. No deadline is included for the development of these methods.
- Up to $500,000 for EPA staff training programs.
- Up to $500,000 for the development and implementation of a Vector Expedited Review Voucher Program which will allow for expedited review of insecticides to help prevent the spread of vector borne diseases. This program is to be implemented within 1 year.
- Up to $500,000 for the Pesticide Surveillance program.
In addition to these “set asides”, EPA will now also be allowed to use application fees for registration review costs during government shutdowns or furloughs.
Bilingual Labeling Requirements
All agricultural and restricted use non-agricultural pesticides must provide Spanish translation of key labeling information either on the container label or via a scannable link on the product label. While antimicrobials and other non-agricultural products, that are not restricted use pesticides, may opt to omit the translation from the label, they will be required to provide a link to the product safety data sheet in Spanish. The deadline for compliance is three years from the date of enactment for all restricted use pesticides (RUP), with longer deadlines for non-RUP products based on their Acute Toxicology Category.
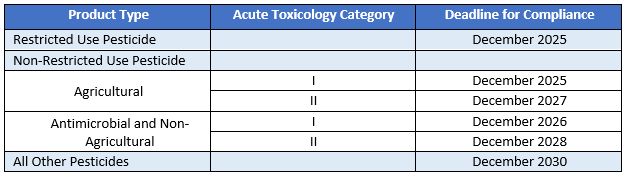
Federally these translations and/or related links and statements can be made using non-notification procedures.
We are pleased to see the continuation of the Fee for Service model though there are increased fees, timelines, and regulatory requirements. SRC will continue to monitor regulatory updates and deadlines and notify our clients of key dates.
Contact SRC with any questions about how PRIA 5 will affect your business.
 Rebecca Mannion
Rebecca Mannion
Federal Assistant Manager – Regulatory
Posted: 1/5/2023


