State Registration
We stay on top of state registration requirements so you don’t have to.
Filing state registrations and renewals on time, keeping labels up to date, and tracking the differences in state regulatory requirements can bog your team down. SRC is a valuable resource to support your project load. We stay on top of the industry changes so when it comes to filing your registrations, you can be sure the most up-to-date forms, regulations, and compliance terms are being considered with ongoing state regulation research and support. Our established relationships with state agencies and our proven track record mean your registrations and renewals are executed efficiently and effectively.

Did You Know?
SRC also has the expertise to manage your Fertilizer and Soil Amendment Registrations.
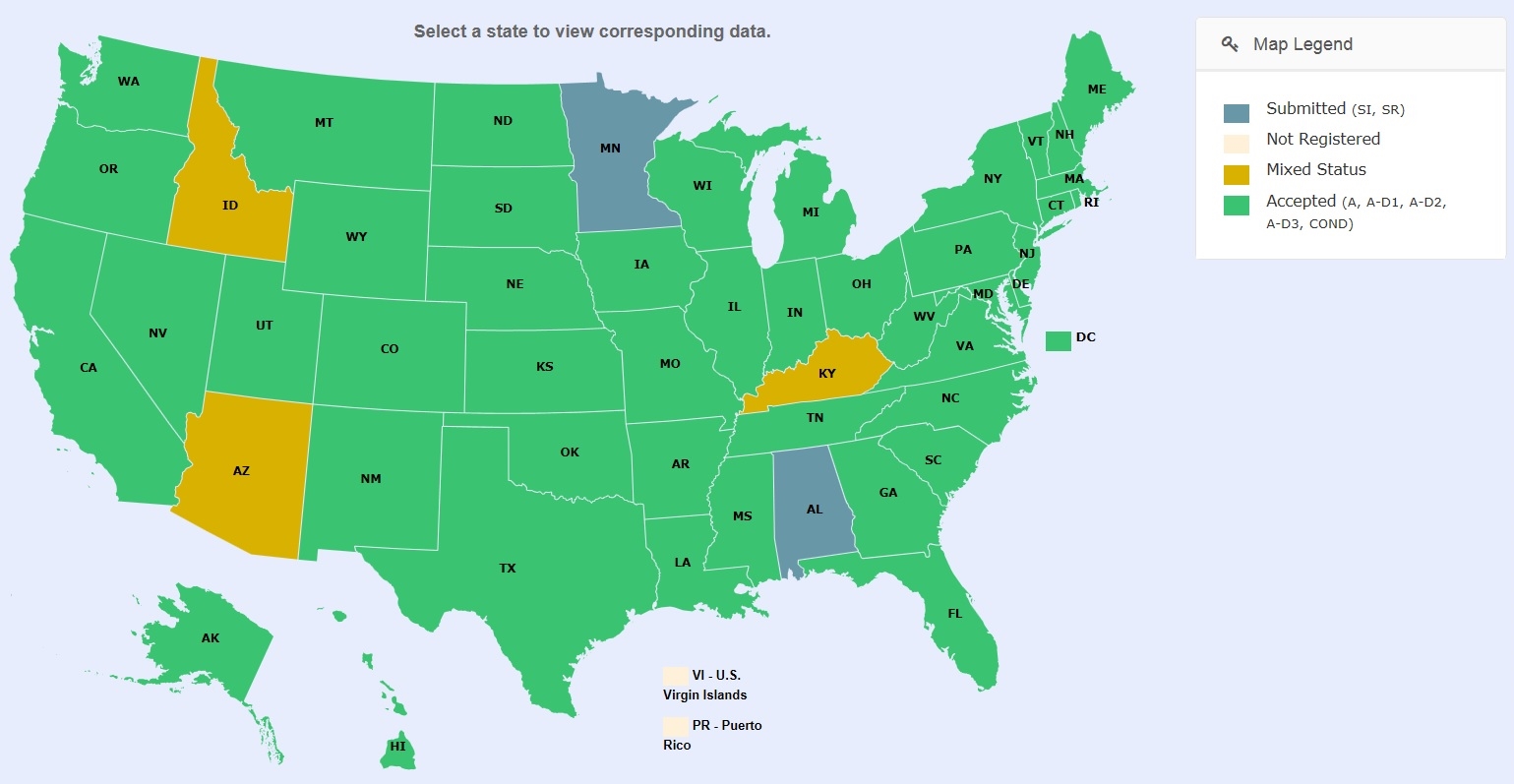
Track your products’ registration status in real time with SRC’s online Service Portal.
SRC represents hundreds of products and manages domestic and international client accounts in the United States and its territories for initial registration, annual renewals, and label updates, as well as other ancillary services. Our expertise includes a variety of product types from 25(b) products to disinfectants, sanitizers, and pesticides. Whether you need to register one product or have numerous products to register in a variety of states, SRC provides comprehensive state registration and compliance services. With our real-time SRCPortal, you have access to track the progress of your registration at any time and on any device!
Product Types
- Household and institutional disinfectants and sanitizers
- Pesticidal devices
- Insecticides
- Pet spot-ons
- Impregnated materials
- Rodenticides
- Multi-packs, co-packs, and kits
- 25(b) products
- Algicides
- Biopesticides
- Fungicides
- Food contact sanitizers
- Adjuvants
- Treated articles
- Antimicrobial pesticide manufacturing products
- High level disinfectants/sterilants
- Towelettes/wipes
- Organic ag input materials
- Organic processing (sanitizers, materials, etc.)
- Fertilizers and soil amendments
Our Services
- Label review
- Initial registration
- Registration renewal and tracking
- Training: state registrations, label review
- Document update submissions
- Input strategy
- DPR submissions
- SRCPortal
- Negotiation
- Enforcement support
- State regulation research and support
Schedule a complimentary call with our team today!

