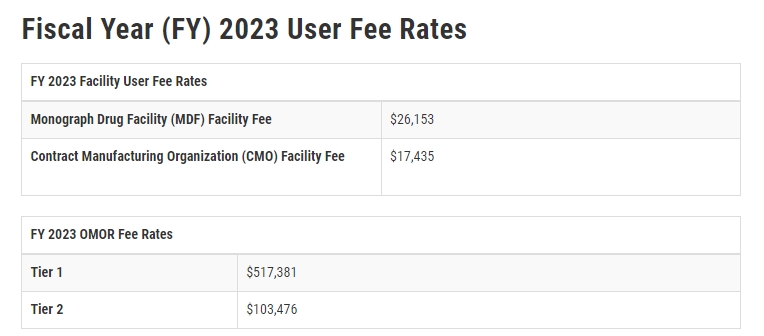
On March 24, 2023, the US Food and Drug Administration (FDA) announced its 2023 fees for OTC monograph drug facilities under the Over-the-Counter Monograph Drug User Fee Program (OMUFA). The Monograph Drug Facility (MDF) fee has increased by $1,975 from $24,178 last year and the Contract Manufacturing Organization (CMO) fee has increased by $1,316 from $16,119 in 2022 (See below chart for current user rates). The official Federal Register Notice was released on March 27, 2023 and details the considerations made when assessing this year’s fee, including reporting facilities operations, June reporting data, and other factors.
 *Screenshot from FDA Website
*Screenshot from FDA Website
OMUFA Facility fees are due on June 1, 2023. FDA will not send an invoice prior to the due date for these fees. To pay your OMUFA fees, follow the instructions on the FDA’s OTC Monograph User Fee Cover Sheet: Step-by-Step Instructions. Payment must be made in U.S. currency by electronic check or wire transfer, payable to the order of the Food and Drug Administration. The preferred payment method is online using electronic check (ACH) or credit card for payments under $25,000 (Discover, VISA, MasterCard, American Express).
Please contact your SRC consultant if you have any questions or would like assistance with payment processing.

Bob MacDonald
VP of Regulatory Services
Posted 03/29/2023


