By Samantha Kappel
Last updated on July 17, 2025
Due to the ongoing Ebola outbreak in Uganda, the United States EPA has engaged its Emerging viral Pathogens Policy (EVP). The current outbreak of Ebola Virus Disease is caused by infection with Sudan ebolavirus. The first confirmed case was identified in a 25-year-old male in the Mubende District of Uganda on September 19th; though it is believed there could have been community spread as early as the beginning of August.
The Center of Disease Control and Prevention (CDC) is working closely with officials in the region to monitor the situation. While the risk of importation and spread to the US is considered low at this time, the CDC has announced an international travel alert to include exit screening for those leaving Uganda and risk assessment questionnaires for air travelers entering the U.S from the affected region. Additionally, the CDC is engaged in domestic preparedness activities including coordination with 10 Regional Special Pathogens Treatment Centers. In response to this outbreak, EPA has triggered its Emerging Viral Pathogens Policy to allow registrants to make indirect, off-label claims against Ebola virus to help arm consumers with important information on proper disinfection to protect against the virus.
Ebola Virus Disease
In humans, Ebola Virus Disease can be caused by 4 different viruses: Zaire ebolavirus, Bundibugyo ebolavirus, Tai Forest ebolavirus, and Sudan ebolavirus. These viruses are typically regional and share the same clinical presentation. While Zaire ebolavirus is the most common etiological agent, the current outbreak is caused by Sudan ebolavirus. Ebola is a zoonotic disease, meaning that the virus can be transferred from animals to humans, with the most likely host reservoir being native bats. Transmission of the virus is spread though direct contact with infected bodily fluids or contaminated surfaces. The virus is not spread by airborne transmission. Following exposure there is a 2 – 21 day incubation period where no symptoms are present, followed by a dry symptom phase and a wet symptom phase during which time the patient is highly contagious. In fatal cases of the disease, death typically occurs 7-10 days following the onset of symptoms. The current outbreak of Sudan ebolavirus is believed to have a mortality rate of approximately 50%. There are currently no FDA approved treatments for infection with Sudan ebolavirus (note that the FDA approved vaccine “Ervebo” is effective against Zaire ebolavirus but not Sudan ebolavirus). There are 2 experimental antibody treatments which have demonstrated some promise in clinical trials, though they are not yet approved. Due to the lack of treatment options, supportive care remains the most effective clinical response.
The Pathway to Make Off-Label Claims Against Sudan ebolavirus
Last week, EPA engaged the Emerging Viral Pathogens Policy (EVP) for off-label claims against the Ebola virus. This policy allows registrants to make indirect, off-label claims against Sudan ebolavirus, if they have previously demonstrated that their product is effective against one or more viruses that are harder to inactivate than the emerging virus. Indirect claims convey to the user that the product is anticipated to be effective against the emerging viral pathogen based on a product’s demonstrated efficacy against viruses similar to the emerging virus.
For example, an indirect, off-label claim may look something like the below tweet:
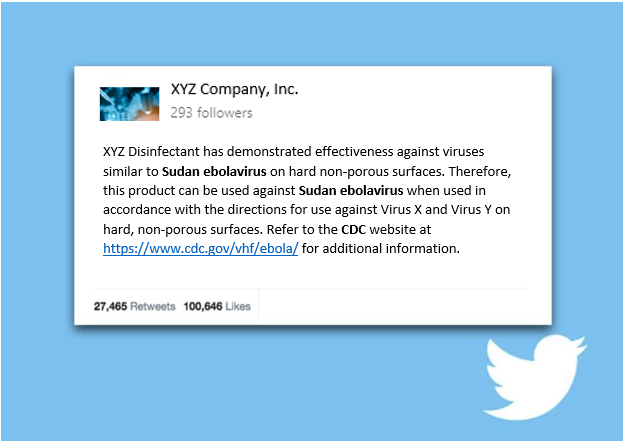
Sudan ebolavirus is an enveloped virus, which is the easiest to kill virus type. To qualify to make indirect, off-label claims against Sudan ebolavirus a registration must have an approved emerging viral pathogen claim against at least one small or one large non-enveloped virus.
Please contact SRC for assistance in making indirect, off-label claims against Sudan ebolavirus.
EPA Releases List L: Disinfectants for Use Against Ebola Virus and Updates List Q: Disinfectants for Emerging Viral Pathogens (EVPs)
EPA is directing consumers to its List L: Disinfectants for Use Against Ebola Virus, where they can find all products that are currently registered for use against Ebola virus. In addition to List L, EPA has added Ebola virus to List Q: Disinfectants for Emerging Viral Pathogens (EVPs) where consumers can search for products approved for use on Tier 1 (enveloped) viruses to find disinfectants that have demonstrated efficacy against viruses that are more difficult to inactivate than Ebola Virus.
If a qualified product is not already included on List Q or L, or if errors are found, registrants may provide their product name and EPA Registration Number to disinfectantlist@epa.gov.
Testing Against Sudan ebolavirus
To make on-label claims against Sudan ebolavirus, efficacy testing must be completed on the virus. To date, no surrogate virus has been approved for testing by EPA. Due to the biosafety level required to work with this virus (BSL-4) it will not be available for testing at most contract laboratories. SRC will continue to monitor this situation and provide updates regarding options for testing and claims against Sudan ebolavirus.
Additional resources and information can be found at the sites included below.
https://www.cdc.gov/vhf/ebola/index.html
https://www.epa.gov/pesticide-registration/list-l-disinfectants-use-against-ebola-virus
https://www.epa.gov/pesticide-registration/disinfectants-emerging-viral-pathogens-evps-list-q